Why e-fuels are more important today than ever
E-fuels are important today as they are produced using renewable energy and drastically reduce the harmful emissions associated with combustion engines. Additionally, e-fuels have a lower total cost of ownership than conventional fuels. E-fuels have several advantages, such as a short refueling process and high energy density, being produced from renewable or decarbonized electricity, being climate neutral in the entire balance and having an unlimited shelf life.
What are e-fuels?
E-fuels are drop-in replacement fuels produced using renewable electricity, water, and CO2 from the air. They are climate neutral in the entire balance and can be used to power vehicles, airplanes and ships. The process uses hydrogen to bind with the CO2 under high pressure using a catalyst.
Excursion: Hydrocarbon
Hydrocarbons are organic compounds composed of hydrogen and carbon atoms. They can be found in petroleum and natural gas and are introduced into the environment through their use as fuels and chemicals. The energy that’s within the hydrocarbon in our fuels is released through combustion. Combustion of hydrocarbons is a chemical reaction where a hydrocarbon reacts with oxygen to create carbon dioxide, water, and heat. Incomplete combustion of hydrocarbons produces the most oxidized form of carbon (carbon dioxide) as a product. Carbon monoxide, a by-product of hydrocarbon combustion, is a primary pollutant in the troposphere. In conclusion burning fossil fuels can have a range of negative side effects, including non-renewability, mercury emission, air pollution and climate change. Carbon dioxide emissions from burning fossil fuels are the main contributor to climate change, leading to a changing climate and trapping heat in the atmosphere. That’s why we are obligated to turn our energy supply away from hydrocarbons of fossil origin towards renewable sources.
First ingredient: green hydrogen
Green hydrogen is produced through electrolysis, a process that splits hydrogen from water using an electric current. The electricity used for this process can be obtained from renewable sources such as on-site solar or wind, making it green. The process of producing green hydrogen results in energy conversion losses of around 30 percent which means that 70 percent of the energy expended is bound in hydrogen. (Source: BMBF 2022)

Green Hydrogen is one of the main ingredients for efuels. The electricity used for this process can be obtained from renewable sources such as on-site solar or wind
Second ingredient: carbon capture
Carbon capture is the process of collecting carbon dioxide (CO2) from a point source, such as a smokestack or gas well, and storing it in an underground facility. There are two main types of carbon capture: post-combustion and pre-combustion. Post-combustion processes use liquid to chemically remove CO2 before it is released into the atmosphere, while pre-combustion processes convert fuel into a gaseous mixture of mostly hydrogen and CO2, which is then separated. CO2 gained from this process is the second important ingredient needed to produce an e-fuel.
The production process of e-fuels
E-fuels are synthetic methanol produced by a complex process using water, hydrogen and carbon dioxide. They are differentiated from biofuels as they are produced from renewable or decarbonised electricity. The production process involves extracting hydrogen through electrolysis of water, then combining it with carbon dioxide to produce the fuel. One often executed process is the Fischer-Tropsch-Synthesis. The Fischer-Tropsch synthesis is a condensation polymerization reaction of CO that produces hydrocarbons, olefins, paraffins and oxygenates. It occurs in the presence of metal catalysts at temperatures of 150–300 °C (302–572 °F) and pressures of one to several tens of atmospheres. E-fuels offer similar performance to petrol and diesel but have a lower environmental impact due to their production process.
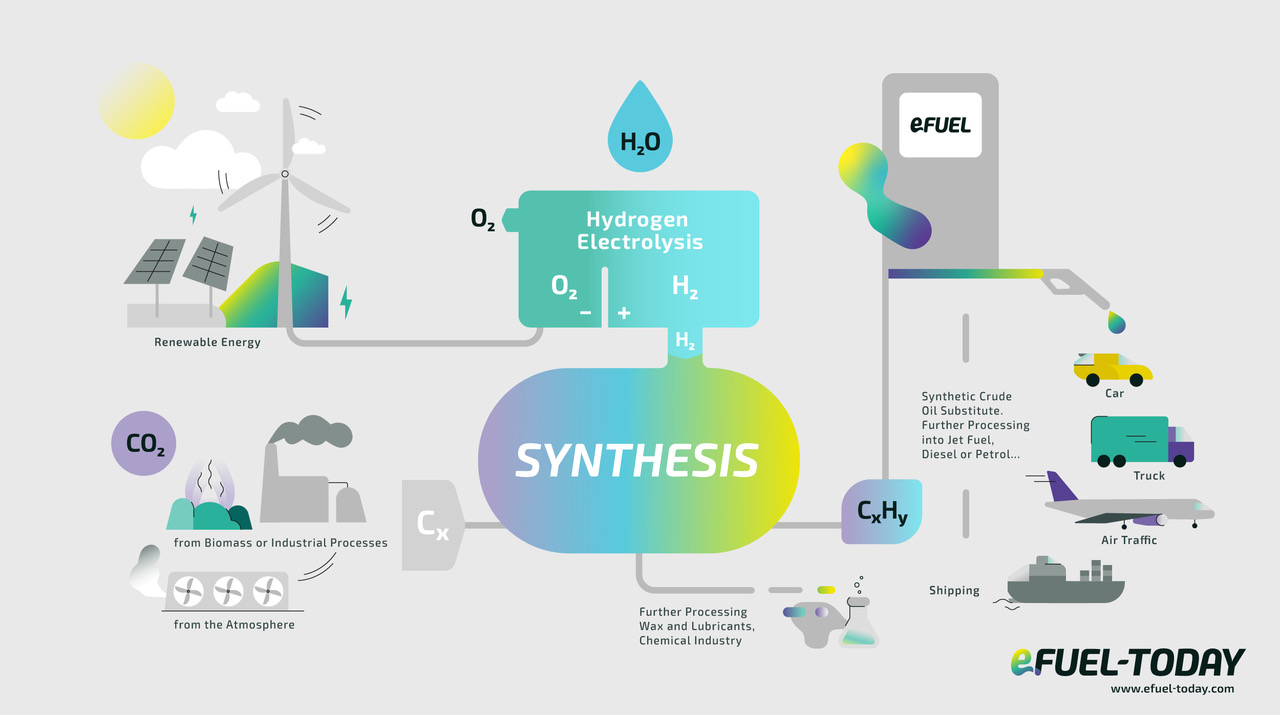
E-fuels are synthetic methanol produced by a complex process using water, hydrogen and carbon dioxide, the production process is illustrated by the diagram.
When can an e-fuel be considered climate neutral
To be climate neutral, e-fuels must be produced from green electricity and have a closed CO2 cycle when viewed from a holistic “well-to-wheel” perspective. Well-to-wheel (WtW) is a measurement used to accurately evaluate and compare vehicle emissions. It calculates the total climate impact of a product or service from its production to its use.
That’s how far the industry is when it comes to the production of e-fuels
Industry mass production of e-fuels is still in its early stages. Porsche has invested over $100 million in the development and production of e-fuels, including $75 million in HIF Global LLC in April 2022 (Source: Porsche 2022)In Germany H&R Refining GmbH and Mabanaft GmbH & Co. KG have signed an agreement to establish the joint venture P2X Europe. The joint venture will purchase carbon-neutral products such as E-Fuels and petrochemical specialties such as waxes from corresponding projects and market them through the respective distribution channels of Mabanaft and H&R. Jonathan Perkins, CEO of Mabanaft, highlights the importance of this cooperation, commenting that it will allow them to bring CO2-neutral E-Fuels to their customers. The project is planned to start producing and marketing synthesis-based e-fuels and waxes from renewable raw materials at the H&R production site in Hamburg starting in September 2022 (Source: Mabanaft 2022)
What are the price predictions for one liter of e-fuel
E-fuels prices are expected to be between EUR 1.22 and EUR 2.17 in 2025, and between EUR 1.45 and EUR 2.50 in 2050. Prices for petrol with an eFuels admixture will cost between EUR 1.34 and EUR 1.36 in 2025. Alternative fuel fleets can obtain significantly lower fuel prices than those reported by entering into contracts directly with local fuel suppliers.
Sources:
BMBF (2022): https://www.bmbf.de/bmbf/shareddocs/kurzmeldungen/de/wissenswertes-zu-gruenem-wasserstoff.html
eFuel alliance (2022): https://www.efuel-alliance.eu/efuels/costs-outlook
Porsche (2022): https://newsroom.porsche.com/en/2022/company/porsche-commitment-industrial-production-efuels-investment-hif-global-llc-27935.html



